|
AHP down on probe talk
|
 |
September 9, 1999: 12:39 p.m. ET
FBI said to investigate FDA over how it approved company's diet drug Redux
|
NEW YORK (CNNfn) - American Home Products Corp. shares declined more than 7 percent Thursday on reports that the Federal Bureau of Investigation is investigating how it received government approval to sell its diet drugs.
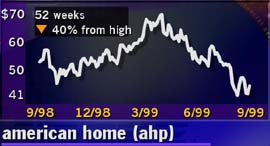
AHP (AHP) shares fell 3-1/16 to 39-1/4 in mid-morning New York trading after several media reports said FBI investigators are questioning the Food and Drug Administration about how regulators concluded AHP's diet drug, Redux, was safe.
FBI officials would not confirm or deny an investigation; officials at the FDA did not return telephone calls.
The reports said investigators are focusing on whether AHP and other companies provided all the information necessary for the FDA to make a proper decision when it approved the company's diet drugs. The drugs -- Redux and its predecessor Pondimin, both part of the "fen-phen" diet drug combination -- were pulled off the shelves at the FDA's insistence in 1997.
The drugs were found to cause heart and lung problems in roughly 30 percent of the patients evaluated by FDA-approved doctors, according to the September 1997 notice of withdrawal. "This is a much higher than expected percentage of abnormal test results," the FDA said at the time. At that time the FDA had received 66 doctor reports of heart valve disease associated with fen-phen.
The latest news for AHP comes at a time when it's already grappling with thousands of lawsuits that could add up to millions of dollars in legal fees and settlement costs for the Madison, N.J.-based drugmaker.
In early August, a Texas jury awarded a former fen-phen user $23.3 million in damages for heart problems. It was the first jury award stemming from individual fen-phen cases, although AHP has disclosed settling about 20 such cases out of court and received particular attention on the CBS news magazine show, "60 Minutes."
Also in August, a federal judge in Philadelphia granted class-action status to a group of former fen-phen users. And AHP's Wyeth-Ayerst Laboratories division agreed recently to settle claims by about 36,000 women who said they were injured by the company's Norplant birth control device.
For its part, AHP denies any knowledge of an investigation, and denies any wrongdoing.
"American Home Products is absolutely unaware of any investigation by the U.S. Department of Justice or the Federal Bureau of Investigation relating to diet drugs," AHP said in a statement issued by its general counsel. "We have made a considerable effort to determine whether such an investigation exists and have been unable to do so. We know of no basis for such an investigation, and we are confident that American Home Products' actions with respect to Pondimin and Redux were at all times lawful and appropriate."
|
|
|
|
|
|
American Home Products
|
Note: Pages will open in a new browser window
External sites are not endorsed by CNNmoney
|
|
|
|
 |

|

