|
J&J down 12% on drug pull
|
 |
March 24, 2000: 4:11 p.m. ET
Withdrawal of heartburn drug triggers downgrades, profit estimate revisions
|
NEW YORK (CNNfn) - Shares of drug and consumer products maker Johnson & Johnson fell about 12 percent Friday after the company said it would stop marketing the popular heartburn medication Propulsid in the United States in the face of fatal side effects in some patients.
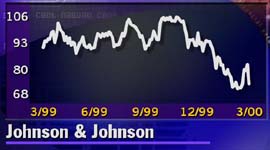
The sell-off of Johnson & Johnson (JNJ: Research, Estimates) stock, one of the 30 companies that make up the Dow Jones industrial average, helped weigh down the index. J&J stock slipped 9-3/8 to 70-5/8 in late afternoon trading. The stock has traded from a low of 66-1/8 to as high as 106-7/8 over the past year.
Wall Street analysts said the news - which comes only a few days after the withdrawal of another big drug, Warner-Lambert Co.'s (WLA: Research, Estimates) diabetes treatment Rezulin - was putting pressure on the rest of the pharmaceutical sector. The American Stock Exchange Pharmaceutical Index, which measures the performance of about a dozen large U.S. and European drug makers, slipped about 3 percent Friday.
A top-selling drug
J&J's Janssen Pharmaceutica unit announced late Thursday it would sharply limit the availability of Propulsid, which has been linked to 341 reports of heart rhythm abnormalities and about 80 deaths since its introduction in 1993. Over the past few years, J&J has warned doctors to be highly cautious in prescribing Propulsid and has changed the drug's labeling, but the company said Thursday that inappropriate use of the drug has continued despite the warnings.
In response to the news, several Wall Street analysts cut their ratings on the company's stock and cut earnings estimates. Propulsid had sales of nearly $1 billion last year and was one of New Brunswick, N.J.-based J&J's best-selling medications. The new restrictions on the drug are expected to bring sales down to about $250 million this year.
While the news is a setback for J&J, the company has taken the lead in issuing warnings about the drug, said Anne Malone, medical supply analyst at Salomon Smith Barney, which rates the company's shares as a "buy."
"It's a pretty substantial drug in J&J's pipeline, but in the grand scheme of things, (it's not that significant for) a company that has $28 billion in revenues to lose $450 million-to-$500 million" in sales, she said. "In the long run, they'll get over this speed bump."
Ratings cut
But other Wall Street analysts were not as forgiving, cutting their ratings on the stock.
J.P. Morgan reduced its rating on Johnson & Johnson to "market performer" from "buy," lowered its 2000 earnings estimate to $3.36 per share, from $3.39, and cut its 2001 estimate to $3.80 per share, from $3.85.
Merrill Lynch also cut its rating on J&J to near-term "accumulate" from "buy," while SG Cowen reduced the stock to "buy" from "strong buy," and cut its 12-to-18 month price target to $92 per share from $110.
J&J said Propulsid will be removed from U.S. pharmacies by July 14, giving patients time to find alternatives. The company and the Food and Drug Administration said they were working to establish a limited access program to keep the drug available for certain patients who have not been helped by other treatments.
The company plans to discuss the drug's future with regulatory agencies in the other 89 countries where it is sold.
Earlier this week, Warner-Lambert pulled Rezulin, a controversial diabetes treatment, from the market. The drug was linked to fatal liver failure in some users. 
-- from staff and wire reports
|
|
|
|
|
|
Johnson & Johnson
|
Note: Pages will open in a new browser window
External sites are not endorsed by CNNmoney
|
|
|
|
 |

|

