|
P&G osteoporosis drug OK
|
 |
April 17, 2000: 4:18 p.m. ET
Procter & Gamble, Aventis approved to co-market new Actonel treatment
|
NEW YORK (CNNfn) - The U.S. Food and Drug Administration has given marketing approval to a quick-acting osteoporosis drug made by Procter & Gamble Co., the company said Monday.
Procter & Gamble (PG: Research, Estimates) will co-market the treatment, called Actonel, with Aventis S.A. (AVE: Research, Estimates) of Germany. Fourteen other countries have indicated they will approve the drug, P&G said.
According to clinical data presented by the companies, Actonel is the first osteoporosis drug to consistently reduce incidents of spinal fractures within one year of treatment. Data shows that about 20 percent of untreated osteoporosis patients who experience a spinal fracture will suffer another fracture within a year, P&G said.
"The approval of Actonel in the U.S. represents a significant milestone for our healthcare business," P&G CEO Durk Jager said in a statement.
 The drug's potential side effects include upper respiratory infection and back and joint pain. The drug's potential side effects include upper respiratory infection and back and joint pain.
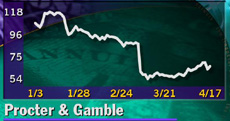
Shares of Cincinnati-based P&G, a component of the Dow Jones industrial average, gained 6 to stand at 69 at 4 p.m. ET, amid a broad rally for U.S. stocks after Friday's steep losses.
American depositary receipts of Aventis added 9/16 to 57-5/16. 
|
|
|
|
|
|
Procter & Gamble
|
Note: Pages will open in a new browser window
External sites are not endorsed by CNNmoney
|
|
|
|
 |

|

