|
Genentech drug trial fails
|
 |
June 16, 2000: 10:24 a.m. ET
Biotech company says heart attack drug wasn't effective in clinical tests
|
NEW YORK (CNNfn) - Biotechnology firm Genentech Inc. disclosed Friday that its experimental heart attack drug failed a key clinical trial, putting the future of the proposed treatment in doubt.
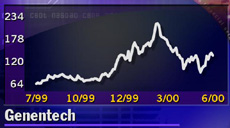
Genentech (DNA: Research, Estimates) stock fell after the news was released, losing 4-1/4 to 133-1/4 in early trading.
 The South San Francisco, Calif.-based company said that preliminary results show that the heart attack treatment, part of a class of biotech drugs called monoclonal antibodies, "did not meet its primary objectives" in a Phase II clinical study of 415 patients. Drugs must successfully complete three phases of human tests before they can be approved. The South San Francisco, Calif.-based company said that preliminary results show that the heart attack treatment, part of a class of biotech drugs called monoclonal antibodies, "did not meet its primary objectives" in a Phase II clinical study of 415 patients. Drugs must successfully complete three phases of human tests before they can be approved.
"We are disappointed in the preliminary analysis of the data," Genentech chief medical officer Susan D. Hellmann said. "However, we plan to meet with our investigators this summer to evaluate the implications of the data, and we remain committed to developing and improving treatments for patients experiencing heart attack."
Genentech's existing products include breast cancer drug Herceptin and Rituxan, a treatment for non-Hodgkin's lymphoma. 
|
|
|
|
|
|
Genentech
|
Note: Pages will open in a new browser window
External sites are not endorsed by CNNmoney
|
|
|
|
 |

|

